Modular Clean Room Panels for GMP Compliance: Complete Guide (2026)
Key Takeaways
- GMP-compliant cleanroom panels must satisfy ISO 14644, EU GMP Annex 1 (2022), and in North America, FDA 21 CFR Part 211
- Surface roughness (Ra value) is the most commonly overlooked specification: ISO 5/6 areas require Ra ≤ 0.8 µm; ISO 7/8 areas require Ra ≤ 1.6 µm [source:fumex]
- Rock wool panels achieve Euroclass A1 fire performance; PIR panels typically achieve B-s1-d0 under EN 13501-1
- Air-tight joint systems reduce HVAC energy load by an estimated 20–30% compared to poorly sealed installations
- GMP-grade panels cost 30–60% more than standard industrial panels — but the cost of a failed audit or contamination event is far higher

1. What Is GMP Compliance for Cleanroom Panels?
In pharmaceutical, biotechnology, and advanced medical device manufacturing, Good Manufacturing Practice (GMP) is the non-negotiable framework ensuring products are consistently produced and controlled to appropriate quality standards. While GMP covers everything from personnel training to documentation, its physical foundation is the cleanroom itself.
GMP compliance for cleanroom panels refers to the specification, installation, and ongoing performance of the wall, ceiling, and door systems forming the primary barrier against contamination. Selecting these panels is far more than a construction choice — it is a critical compliance decision with direct regulatory implications.
Panels must withstand aggressive cleaning regimens, prevent particle generation, and maintain strict pressure differentials. Three standards form the governing framework:
- ISO 14644 Series: The global benchmark for cleanrooms and associated controlled environments. Parts 1 (classification), 3 (testing), and 4 (design and construction) are directly relevant to panel specification.
- EU GMP Annex 1 (2022 Revision): This pivotal guideline for sterile medicinal products became effective September 2022 and places unprecedented emphasis on Contamination Control Strategy (CCS). It explicitly states: "In cleanrooms and critical zones, all exposed surfaces should be smooth, impervious and unbroken in order to minimize the shedding or accumulation of particles or micro-organisms."
- FDA 21 CFR Part 211: The U.S. regulation for finished pharmaceuticals mandates that "floors, walls, and ceilings of smooth, hard surfaces that are easily cleanable."
The consequences of non-compliance are severe. Regulatory inspections can result in FDA Form 483 observations, warning letters, or consent decrees halting production. More critically, inadequate panels can cause contamination events leading to batch failures, product recalls, and reputational damage. Investing in correctly specified panels from the outset is the most effective risk mitigation strategy available.
2. Cleanroom Classification & Panel Requirements
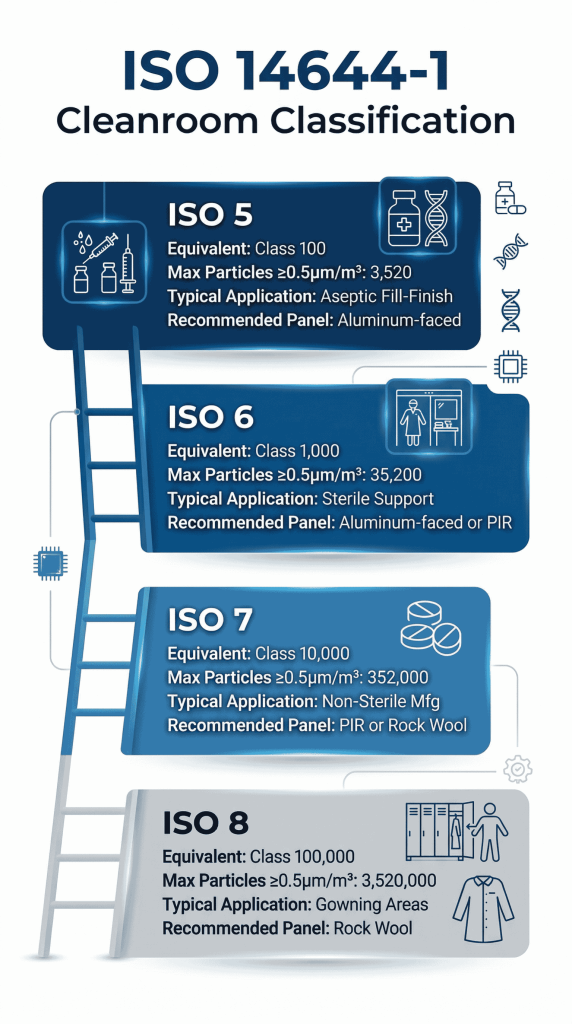
The cornerstone of any cleanroom design is its ISO classification, defined by ISO 14644-1:2015. This standard classifies air cleanliness by the concentration of airborne particles ≥ 0.5 µm per cubic meter. The lower the ISO class number, the cleaner the environment.[Source:GOTOPAC]
For pharmaceutical manufacturing, typical classifications range from:[Source:Setra]
- ISO 5 (equivalent to former Class 100): Critical aseptic processing areas — fill-finish lines, sterility testing
- ISO 6 (equivalent to former Class 1,000): Background environments for ISO 5 areas; less critical sterile preparation
- ISO 7 (equivalent to former Class 10,000): Preparation and support areas for sterile products
- ISO 8 (equivalent to former Class 100,000): Less critical non-sterile manufacturing; gowning areas
The 2022 revision of EU GMP Annex 1 further tightened expectations for surface quality, explicitly requiring that surfaces be "smooth, impervious and unbroken." EU GMP Annex 1 also uses a parallel Grade A–D system that aligns with ISO 14644-1 classification.
ISO Class, Application, and Panel Requirements
| ISO Class | Typical Application | Recommended Panel Material | Surface Roughness (Ra max) | Air Tightness |
|---|
Particle count limits per m³ (≥ 0.5 µm) per ISO 14644-1:2015: ISO 5: 3,520; ISO 6: 35,200; ISO 7: 352,000; ISO 8: 3,520,000.
3. Panel Material Selection for GMP Cleanrooms

The core material of a modular cleanroom panel defines its fundamental performance characteristics: fire safety, thermal insulation, and structural integrity.
1. Rock Wool (Mineral Wool) Core Panels: Rock wool panels offer exceptional fire resistance, typically achieving Euroclass A1 or A2 ratings. They are a robust, cost-effective solution for ISO 7 and ISO 8 areas, corridors, and external walls where fire performance is the primary driver.
2. PIR/PUR Core Panels: These foam-core panels provide superior R-value per unit thickness (R-7 to R-8 per inch), leading to thinner wall profiles for the same insulation performance. Suitable for ISO 6 through ISO 8 applications.
3. Aluminum-Faced Panels: For the most critical environments (ISO 5 and ISO 6), aluminum surfaces are inherently non-porous, highly chemical-resistant, and can be finished to an ultra-low Ra value (<0.5 µm).
GMP Cleanroom Panel Material Comparison
| Material | Fire Rating | R-value per 25mm | Chemical Resistance | Surface Ra | Best ISO Class | Cost |
| Rock Wool Core | A1 (Non-combustible) | 1.1 - 1.3 | Good | 1.6 - 2.0 µm | ISO 7, ISO 8 | $ |
| PIR/PUR Foam | B-s1,d0 / Class A | 1.8 - 2.2 | Very Good | 0.8 - 1.6 µm | ISO 6, ISO 7, ISO 8 | $$ |
| Aluminum-Faced | Varies (A to B) | 0.9 - 2.0 | Excellent | < 0.5 - 0.8 µm | ISO 5, ISO 6 | $$$ |
Selection Guidance: The choice often starts with fire code and moves to cleanliness. For sterile areas, the impervious surface of aluminum-faced panels is paramount. For general GMP production, PIR panels offer the best balance. We've found that specifying rock wool for perimeter walls and PIR for internal partitions is a common and compliant hybrid approach.
4. Surface Science: What Makes a Panel “GMP-Grade”?
Ra (arithmetic average roughness) measures the average deviation of a surface from a true flat plane. Lower Ra values indicate smoother surfaces — which are easier to clean, less likely to harbor microorganisms, and better at resisting chemical attack.
EU GMP Annex 1 (2022) requires that cleanroom surfaces be "smooth, impervious and unbroken." In practical specification terms, this translates to:
- ISO 5/6 critical zones: Ra ≤ 0.8 µm
- ISO 7/8 support areas: Ra ≤ 1.6 µm
- Standard industrial panels (non-GMP): Ra can be 3.0 µm or higher — unacceptable for GMP environments
The ideal cleaning condition is achieved at Ra values between 0.25 and 0.60 µm according to published surface finishing guidance, representing the point at which cleaning agents make optimal contact with the surface without becoming trapped in surface depressions. Standard industrial coated steel panels almost never meet this threshold without specific GMP surface treatment.
Non-Shedding Performance
Verified through wipe tests per IEST-RP-CC018 (Institute of Environmental Sciences and Technology Recommended Practice) and continuous particle count monitoring. A non-shedding surface is required to maintain ISO class certification during routine operations. Any surface that generates particles under mechanical contact, thermal cycling, or chemical exposure is non-compliant.
Chemical Resistance
GMP cleanrooms routinely use isopropyl alcohol (IPA), sodium hypochlorite (bleach), hydrogen peroxide, and peracetic acid as cleaning and disinfecting agents. GMP-grade panel surfaces must withstand repeated exposure to these agents without fading, blistering, delamination, or surface degradation. Suppliers should provide chemical resistance test data covering the specific agents used in the facility.
Biostatic Properties
Many modern GMP panel systems incorporate antimicrobial additives into the surface coating, inhibiting the growth of microorganisms on the panel surface between cleaning cycles. This is particularly relevant for higher-humidity environments such as oral solid dose manufacturing and food processing cleanrooms.
5. Air Tightness & Pressure Control
A cleanroom's pressure cascade is its dynamic defense against cross-contamination. Positive pressure in cleaner zones prevents uncontrolled airflow from less-clean adjacent areas. Maintaining defined pressure differentials requires both an effective HVAC system and an airtight building envelope — and the panel joint system is the most vulnerable point in that envelope.

Joint System Technologies
Modern GMP cleanroom panel systems use three main joint designs:
- Tongue-and-groove profiles: Provide a mechanically aligned, close-tolerance joint suitable for ISO 7 and ISO 8 applications
- Integrated gaskets (EPDM or silicone): Add a compressible seal layer that accommodates minor surface irregularities and thermal movement
- Cam-lock fastening systems: Provide positive mechanical clamping force that compresses the gasket to a defined pressure, delivering the highest air-tightness performance; recommended for ISO 5 and ISO 6 applications
Air Tightness Testing
Pressure decay testing quantifies the panel system's performance by pressurizing the cleanroom to a defined level, isolating it from the HVAC system, and measuring the rate of pressure loss over time. A well-sealed envelope for ISO 5/6 should achieve pressure decay below 0.25%/hour.
The energy impact of air tightness is significant. A well-sealed building envelope reduces HVAC energy load by an estimated 20–30% compared to poorly sealed installations, because the HVAC system does not have to continuously compensate for uncontrolled leakage. Poor sealing also destabilizes room pressure differentials, forcing HVAC systems to operate at higher fan speeds, shortening filter service life, and increasing maintenance frequency.
6. Application-Specific Cleanroom Panel Requirements
Different end-use applications have specific ISO classification requirements and corresponding panel specifications. The table below covers the most common application types:
| Application | ISO Class | Recommended Panel | Key Requirement | Est. Cost (USD/m²) |
| Pharma (Oral Solids) | ISO 7/8 | Rock Wool, Vinyl-coated Steel | Chemical resistance, Dust-tight | $40 - $65 |
| Biotech / Sterile Fill | ISO 5/6 | PIR, Anodized Aluminum | Ultra-smooth (Ra < 0.8µm), Sterilizable | $70 - $120 |
| Hospital Operating Theater | ISO 5/7 | PIR/Rock Wool, Antibacterial Coated | Anti-static, VHP compatible | $65 - $100 |
| Electronics Manufacturing | ISO 4/5 | PIR, ESD Powder-coated Steel | Static dissipation, Humidity stable | $60 - $95 |
| Food Processing (High Care) | ISO 6/7 | PIR, Stainless Steel 304/316 | Hygienic design, Washdown (IP66) | $80 - $130 |
Cost estimates are indicative benchmarks for 2026 and vary by panel thickness, joint system, surface specification, and project region. GMP panels typically cost 30–60% more than standard industrial panels of equivalent dimensions
7. Five Common Mistakes When Specifying Cleanroom Panels for GMP
1. Ignoring Surface Roughness Requirements — Specify maximum Ra value and require manufacturer's certificate of compliance.
2. Mismatching Panel Material to Cleanroom Class — Follow a zoning approach; use application-specific panels for each ISO class.
3. Underestimating Joint Sealing — Insist on engineered joint systems (tongue-and-groove with gaskets or cam-locks).
4. Not Considering Chemical Resistance — Request chemical resistance test reports for your specific cleaning agents.
5. Selecting Non-Fire-Rated Core Materials — Specify EN 13501-1 classification; demand certification for the complete panel.
8. GMP Cleanroom Panel Inspection Checklist
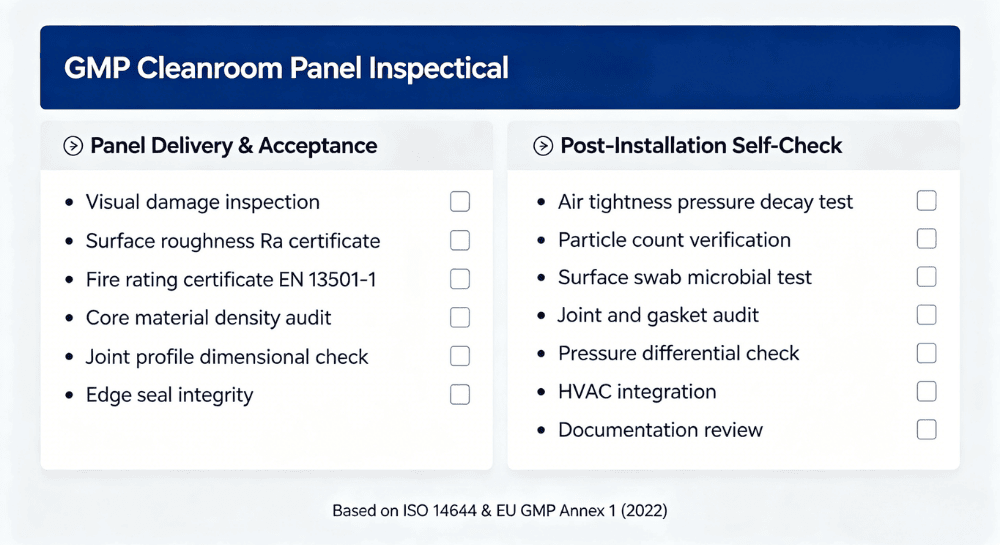
Panel Delivery and Acceptance
Before installation begins, every panel delivery should be verified against the following:
- Visual inspection for transport damage: scratches, dents, surface coating integrity
- Surface roughness verification: supplier Ra certificate plus profilometer spot-check on representative panels
- Fire rating certificate: EN 13501-1 or ASTM E84 — confirm the tested product configuration matches the delivered specification
- Core material audit: verify density (kg/m³) and core type against order confirmation
- Joint profile dimensional accuracy: confirm tongue-and-groove or cam-lock dimensions are within tolerance
- Edge sealing integrity: confirm all factory-applied edge seals are undamaged
Post-Installation GMP Self-Assessment
After installation and before operational qualification, verify the following:
Documentation review: batch numbers, test certificates, installation records, and as-built drawings
Air tightness test: pressure decay or smoke visualization at all joints
Particle count verification at simulated operating conditions
Surface swab test: microbial baseline sampling at representative locations
Joint and seal audit: confirm gaskets are fully compressed along all seam lengths
Pressure differential check: verify cascade pressures are stable at design setpoints
HVAC integration verification: airflow and pressure confirmation with test equipment
9. Frequently Asked Questions
Q: What ISO standard covers cleanroom panels?
The primary reference is the ISO 14644 series. Part 1 (ISO 14644-1:2015) defines classification by airborne particle concentration; Part 4 addresses design, construction, and start-up of cleanroom facilities. For pharmaceutical applications, EU GMP Annex 1 (2022) is the additional governing document.
Q: What is the minimum surface roughness for GMP cleanroom walls?
For ISO 5/6 sterile zones: Ra ≤ 0.8 µm. For ISO 7/8 support areas: Ra ≤ 1.6 µm. Standard industrial coated panels typically have Ra values of 3.0 µm or higher, which do not meet GMP requirements.
Q: Can the same panel type cover both ISO 5 and ISO 8 rooms?
Generally, no. ISO 5 aseptic zones require anodized aluminum or premium-finish surfaces. ISO 8 gowning and support areas can use standard coated steel panels. Using ISO 5-grade panels throughout is compliant but unnecessary and significantly more expensive.
Q: What core material is best for pharmaceutical cleanrooms?
Rock wool for ISO 7/8 perimeter and fire-rated walls (A1 fire rating under EN 13501-1). PIR with aluminum face for ISO 5/6 sterile zones where smoothness, insulation performance, and sterilizability are the priority.
Q: How do you test panel air tightness?
Pressure decay testing: pressurize the room to a defined level, isolate it from the HVAC system, and measure pressure drop over a fixed time period. Smoke visualization at joints is used to identify specific leak locations.
Q: Are cleanroom panels fire-rated?
Yes — fire rating is mandatory for GMP environments. The applicable standard depends on the project market: EN 13501-1 (A1/A2 minimum for pharma) in Europe and many export markets; ASTM E84 Class A in North America. Most pharmaceutical GMP facilities require a minimum of A2 (EN) or Class A (ASTM).
Q: What is the cost difference between GMP and standard panels?
GMP panels cost approximately 30–60% more than standard industrial panels of equivalent dimensions: roughly $40–120/m² for GMP-grade versus $25–50/m² for standard industrial specification. The premium reflects surface treatment, joint engineering, and third-party certification costs.
Q: Can modular cleanroom panels be relocated?
Yes. Cam-lock and tongue-and-groove systems are designed for disassembly and reinstallation with minimal material waste. This is a significant operational advantage for pharmaceutical manufacturers who need to reconfigure production areas to accommodate new product lines or process changes.
Q: How long do GMP cleanroom panels last?
With proper maintenance and cleaning protocols, 15–25 years of serviceable life is typical. Replacement is indicated when surface integrity degrades below the specified Ra value, when the core is physically compromised, or when the panel system can no longer maintain the required pressure differentials.
Q: What maintenance schedule do GMP cleanroom panels require?
A structured schedule aligned with GMP documentation requirements: daily visual inspection by operations staff; monthly joint and seal inspection; quarterly surface assessment with Ra spot-check; annual comprehensive audit including air tightness testing, particle counts, and documentation review.





